- Home
- About
- Industrial Layout
- Products
- R&D
-
HR
- Talent Concepts
- Talent Development
Human Resources

- IR
 Products
Products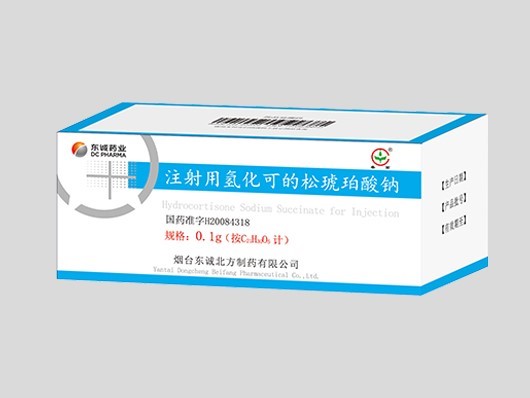
【English name】 Hydrocortisone Sodium Succinate for Injection
【Dosage form】 Freeze dried powder injection
【Approval Number】 National Medical Products Administration Approval Number H20084318
【Specification】 0.1g (calculated as C21H30O5)
【Packing】 10 bottles/box
【Indication】 Used for rescuing critically ill patients, such as toxic infections, anaphylactic shock, severe adrenal insufficiency, connective tissue disease,
severe bronchial asthma and other allergic diseases, and can also be used for the prevention and treatment of acute transplant rejection.